Safety and compliance are paramount in the highly regulated world of medical devices. ISOtrain LMS offers a comprehensive solution to optimize training and compliance processes, solving many of the challenges medical device manufacturers face.
From ensuring regulatory compliance to enhancing product quality and safety, ISOtrain LMS empowers medical device companies to deliver innovative solutions that improve patient outcomes.


Choosing a training solution for your organization goes beyond empowering your team to perform at its best. You need a reliable LMS for medical products that keeps you compliant and competitive. ISOtrain LMS is your trusted solution that understands your needs, whether it’s protecting patients, ensuring compliance or keeping everyone on the same page.
Softek Export LLC is your partner for compliance-focused LMS solutions trusted by medical device leaders. We manufacture products under the highest standards, creating systems that meet your operational needs while ensuring regulatory compliance every time.


We’re more than just a platform. We’re your reliable partner. With decades of experience, you’re getting software and a dedicated partner in training success. We build trust by demonstrating expertise in compliance and industry-specific needs.






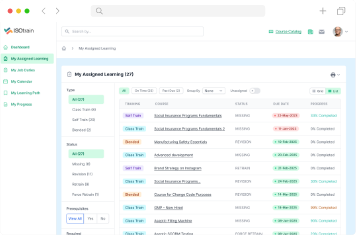


ISOtrain LMS is the cornerstone for medical device manufacturers striving for excellence. By prioritizing safety, quality, and compliance, our solution empowers organizations to navigate regulatory complexities with confidence. Together, let’s shape the future of healthcare innovation while upholding the highest standards of patient care. Choose ISOtrain LMS as your trusted partner on this journey.