At Softek Export, we create validated Learning Management Systems (LMS) for regulated industries. Our LMS, ISOtrain, improves how regulated companies organize, schedule and manage the employee training process. ISOtrain is fully compliant with both company and regulatory requirements, making it the ideal choice for companies working in pharmaceutical, food and beverage, construction, government and other regulated industries.


The training requirements for these industries are complex and changeable. Ensuring you and your team meet the most current compliance standards is essential to avoid warning letters and costly fines. Time-sensitive compliances can be challenging to juggle alongside product updates and staff management, but ISOtrain can solve this. We keep your company compliant with corporate policies and regulatory agencies regardless of what changes your business experiences.
Softek Export LLC is a privately held company headquartered in Miami, Florida. Softek is an agile organization that has developed software and services for a variety of applications like: Training Management, SOP Circulation/Approval, Project Cost Tracking, Direct Labor Distribution, Manufacturing Control, and Instrument Calibration.
We use our extensive experience and knowledge to provide various regulated industries with efficient, versatile and validated training software configured to their specifications. ISOtrain allows companies to stay on top of employee training requirements with ease.
We service various regulated industries to enable them to stay compliant in their training, including:
ISOtrain has been a global LMS leader for over 25 years. Our robust, scalable and secure software includes extensive reporting options. We also maintain an in-house Quality Assurance Team for validation and follow a strict Software Development Lifecycle (SDLC). To ensure ISOtrain meets the highest quality standards, we are also accustomed to hosting vendor audits.
We’re the leading choice for LMS in heavily regulated industries. ISOtrain has been in use for longer than any other product in the market, and we’ve provided countless companies with the resources to train personnel, track training data and analyze statistics. What’s more, ISOtrain provides data tracking so that you can stay compliant in the event of a regulatory audit.
By maintaining complete electronic LMS transactions, you can quickly provide auditors with their requested information. If your organization purchases another company, we can convert the purchased company’s training records from your LMS.
In addition to these many benefits, the main features of ISOtrain include:
Manage the entire lifecycle of your training program from one easy-to-navigate central point of command. Assign specific courses to employees, monitor their progress and track their qualifications. You can also use this portal to instantly notify employees about any changes to compliance requirements.
Our Android and iOS mobile features allow you to use our LMS wherever you are. Control the reporting, training, qualifications and assessment functions entirely remotely.
Our flexible software allows you to add or remove features as your company’s business needs evolve.
Our Quality Assurance (QA) department executes software testing and validation for continual product improvement.
Our LMS makes it easy to stay on top of changing compliance requirements and regulations. The automated system allows your company to standardize training procedures and update educational material.

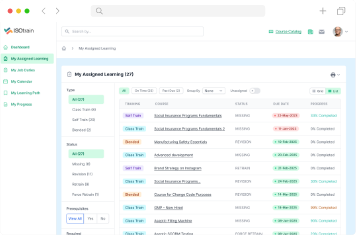


Regulated industries require a specialized LMS to streamline the employee training process. ISOtrain is a Learning Management System that enables your employees to comply with corporate and regulatory requirements. Visit our solutions page today to learn more about our products, or contact us for a free software demonstration.